What Is the Concentration of Oh Ions in Pure Water
Given at 100 C Kw 49 10 14 H301 10105 M. As H3O equals OH- then pH must equal pOH because these are just the negative logs of the respective concentrations which are equal.

The Ionic Product Of Water Is 10 14 What Is The H Ion Concentration In 0 1 M Naoh Solution
Note that this is slightly higher than Kw 1014 at 298 K and given that this is a bond breaking reaction this is perhaps reasonable.

. OH-70 10-5 M Given at 100 C Kw 49 10 14 H301 10105 M. Course Title CHEM 1211. Thus K w OH x H 3 O 10 -7 M x 10 -7 M 10 -14 Notice that K w has no units.
Subscribe and get access to thousands of top quality interact. O ions in pure water is 10 6mol dm 3. The hydrogen ion concentration equals the hydroxide ion concentration and both equal 1 X 10-7 M.
OH1-70 107 M H3O- 7010-9 M. In pure water the hydrogen ion concentration H equals the hydroxide ion concentration OH-. From the equation H 3OH O 147 1014.
This organism would be classified as a _________ In pure water at 25C the concentration of H3O is equal to the concentration of OH-. Here you will find curriculum-based online educational resources for Chemistry for all grades. In pure water at 25C the H3O and OH- ion concentrations are 10 x 10-7 M.
However it grows best at 20 degrees Celsius. The pOH of the solution can be used to calculate the hydroxide ion concentration in asolution of OH- mol L-1 if a hV-concentration is present. Pure water contains water molecules H20 hydronium ions H and hydroxide ions OH-.
Pure water partially breaks down into charged particles in a process called C. In pure water at 25C the H 3 O and OH- ion concentrations are 10 x 10-7 M. 10-7 H30 What is the concentration of OH- ions in pure water.
The value of Kw at 25C is therefore 10 x 10-14. The hydrogen ion concentration equals the hydroxide ion concentration and both equal 1 X 10-7 M. The corresponding concentration of OH ions will be.
What is H0 and OH ion concentration of pure water at 100C. Although K w is defined in terms of the dissociation of water this equilibrium constant expression is equally valid for solutions of acids and bases dissolved in water. What is the concentration of oh ions in pure water a.
In pure water the concentrations of hydrogen and hydroxide ions are equal to one another. 2H 2Ol H 3O H O where the ion product Kw 147 1014 at 303 K. An organism displays some growth at 4 degrees Celsius and at 25 degrees Celsius.
A neutral solution is one that is neither acidic nor basic. The pH of pure water is 7 the negative logarithm of 1 X 10-7. OOH is classified as a negative logarithm to base.
What is it called when the number of hydronium ions and hydroxide ions are equal. Self-ionization What is the concentration of H3O ions in pure water. 6767 B 0465 C 7000 D 7233 E 8446 12 What.
Addition of H and OH ions concentration in pure water at 90C is - 1 10-14 2 10-12 32 x 10600 4 2 x. In pure water at 25C the H 3 O and OH ion concentrations are 10 x 10 7 M. 0 x 10 -7.
Does Pure water have more H. Pure water is considered to neutral and the hydronium ion concentration is 10 x 10-7 molL which is equal to the hydroxide ion concentration. OH1-70 107 M H3O- 7010-9 M.
In pure water at any temperature H3O equals OH-. Pure water contains water molecules H20 hydronium ions H and hydroxide ions OH-. For simplicity hydronium ions are referred to as hydrogen ions H.
A pure beverage with no ions is 25C at a concentration of HO and OH- 1 respectively. Click hereto get an answer to your question 28. In pure water there are an equal number of hydrogen ions and hydroxide.
Chemistry questions and answers. 10 What is the concentration of hydroxide ions in pure water at 300C if Kw at this temperature is 147 10-14 A 100 x 10-7 M B 130 x 10-7 M 147 x 10-7 M 893 x 10-8 M 121 x 10-7 M 11 What is the pH of pure water at 400C if the Kw at this temperature is 292 10-14. At 0 C pKw 149.
The concentration of both hydronium ions and hydroxide ions is 1. The pH of pure water is 7 the negative logarithm of 1 X 10-7. In pure water the hydrogen ion concentration H equals the hydroxide ion concentration OH-.
Given that this is a neutral solution H 3O H O. PKw pH pOH. The number of H3O and OH- ions formed by the ionisation of pure water must be equal from the equation.
The concentration of both hydronium ions and hydroxide ions is 1. Pages 7 This preview shows page 2 - 4 out of 7 pages. A neutral solution is one that is neither acidic nor basic.
What is the concentration of OH ions in pure water a 10 7 M c 554 M b 07 M d 10. Although K w is defined in terms of the dissociation of water this equilibrium constant expression is equally valid for solutions of acids and bases dissolved in water. Furthermore what is the concentration of hydronium ions in water at 25 C.
Concentration of H 3 O is the concentration of H. The value of K w at 25C is therefore 10 x 10-14. Is H3O equal to Oh.
The value of K w at 25C is therefore 10 x 10 14. 10-7 OH- What is the product of the H3O ion and the OH- ion concentrations in water. What is the concentration of hydrogen ions and hydroxyl ions in pure water.
Ionization and pH In fact the concentrations of H and OH ions in pure water at 25C are 107 mol l1 an extremely small value when compared. Pure water or any other aqueous solution in which this ratio holds is said to be neutral. Calculate the concentrations of H3O and OH ions in a 0050 M Ba OH2 solution.
What Is The Concentration Of Hydroxide Ions In Water. A 10 14mol dm 3 B 10 8mol dm 3 C 10 6mol dm 3 D 10 7mol dm 3 Easy Solution Verified by Toppr Correct option is C In pure water concentration of H ions are always equal to OH. Since we know that one liter of pure water at 25C contains 10 x 10 -7 moles of hydronium H 3 O ions per liter of pure water and exactly the same number of hydroxide OH ions we can calculate the value of K w.

The Ph Of Pure Water At 50 C Is 6 63 What Is The Value Of K W At 50 C Youtube

Pin By Xxxeena On Apuntes Study Notes Study Motivation School Organization Notes

Solved 10 What Is The Concentration Of Hydroxide Ions In Chegg Com

The Ionic Product Of Water Is 10 14 What Is The H Ion Concentration In 0 1 M Naoh Solution

Equilib Reactions Ph It All Starts With Water Science Rules Reactions Pure Water
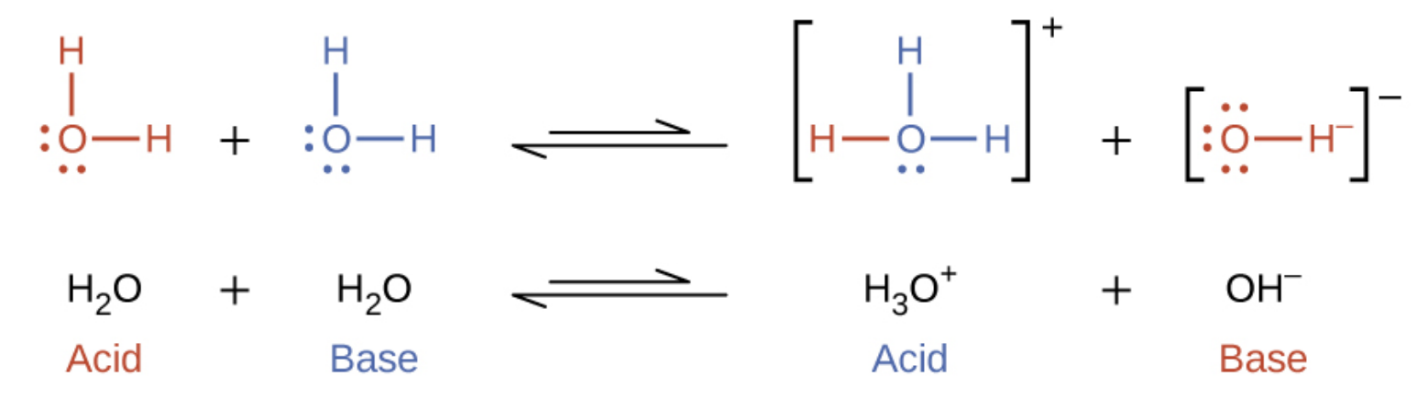
5 2 Autoionization Of Water Ph Poh General Chemistry For Gee Gees

How To Calculate Hydroxide Ion Oh Concentration From Ph Youtube

Conductivity Of Water Dewwool Learn Physics Water Chemical Reactions
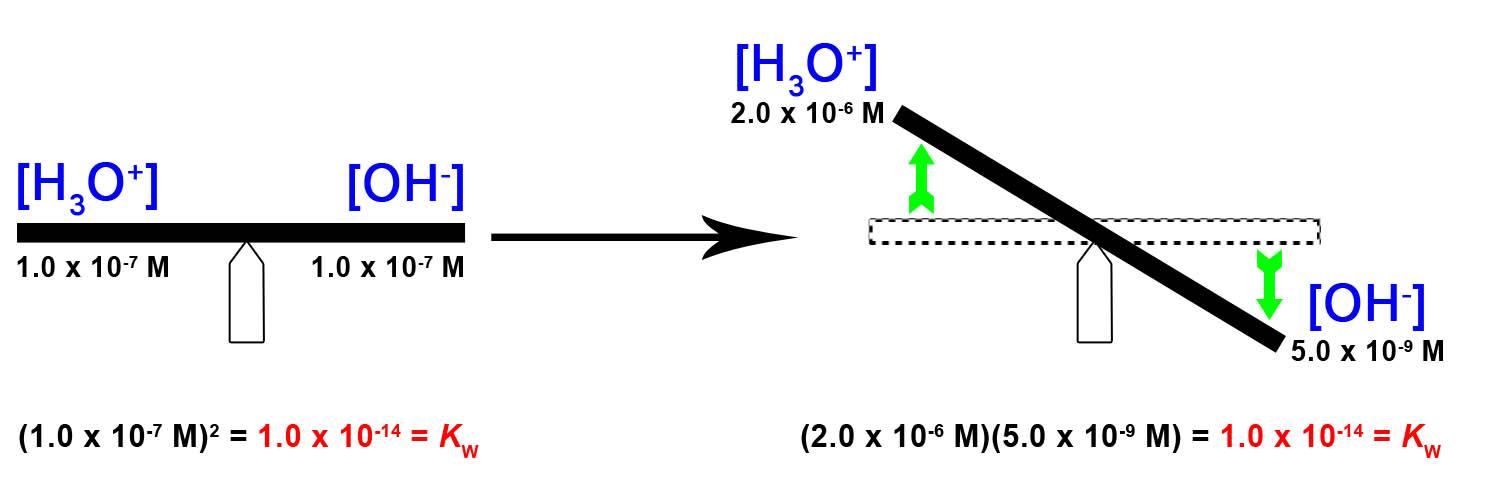
5 2 Autoionization Of Water Ph Poh General Chemistry For Gee Gees
Concentration Of Hydrogen Ions Horiba
When An Acid Is Mixed With Water The Concentration Of H Ion Increases The Ionic Product Of Water Has To Remain Constant So The Concentration Of Oh Ion Decreases Where Do Oh

Is Ph The Measurement Of Hydrogen Ion Concentration Or Ion Activity

Pin By Mike Jones On Aquarium H3bo3 Fluoride Borate

After Completing This Topic You Should Be Able To Describe Effect Of Dilution On The Ph Of An Acid Or Alkali Is Explained In Terms Of The Decreasing Ppt Download
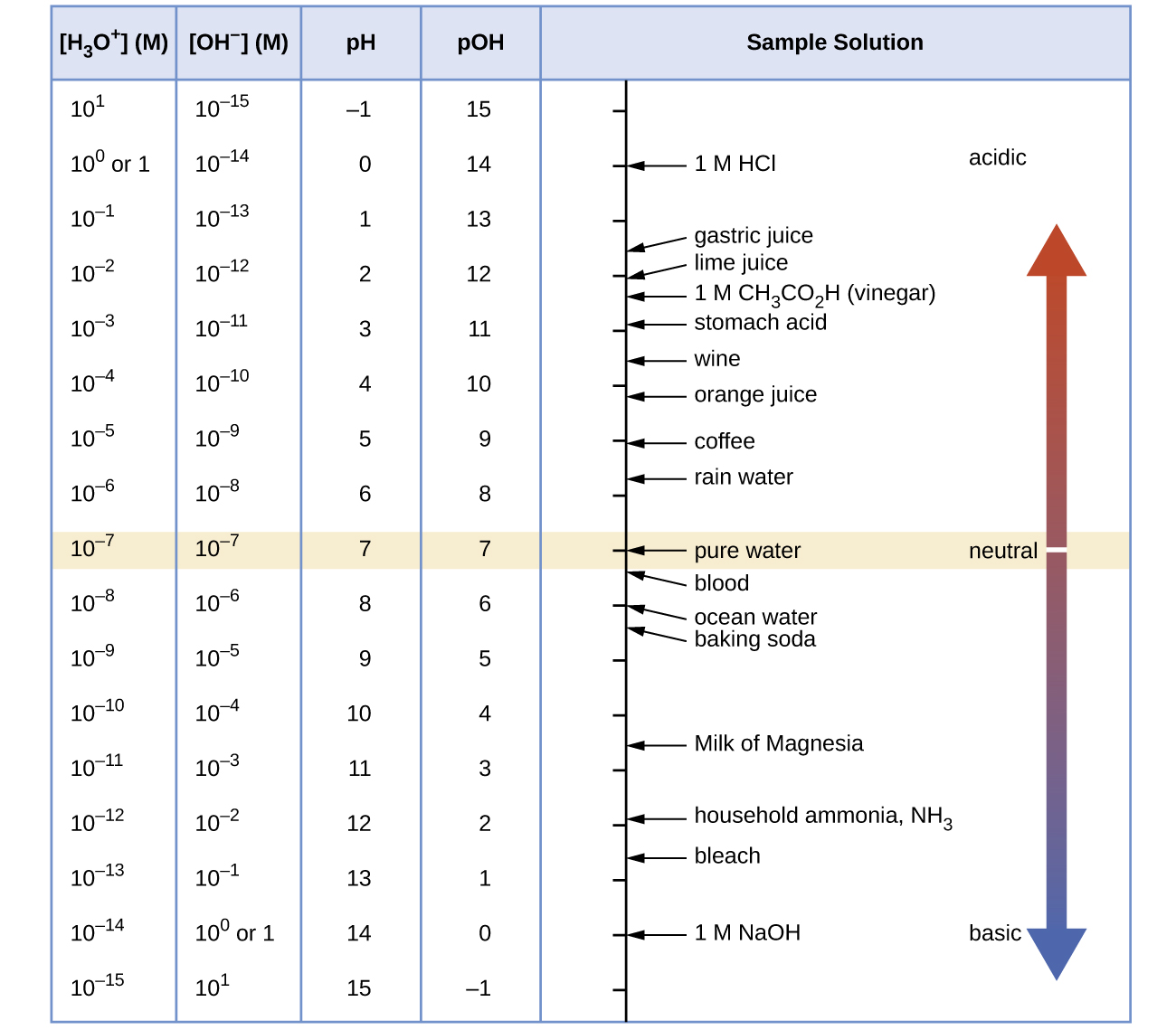



Comments
Post a Comment